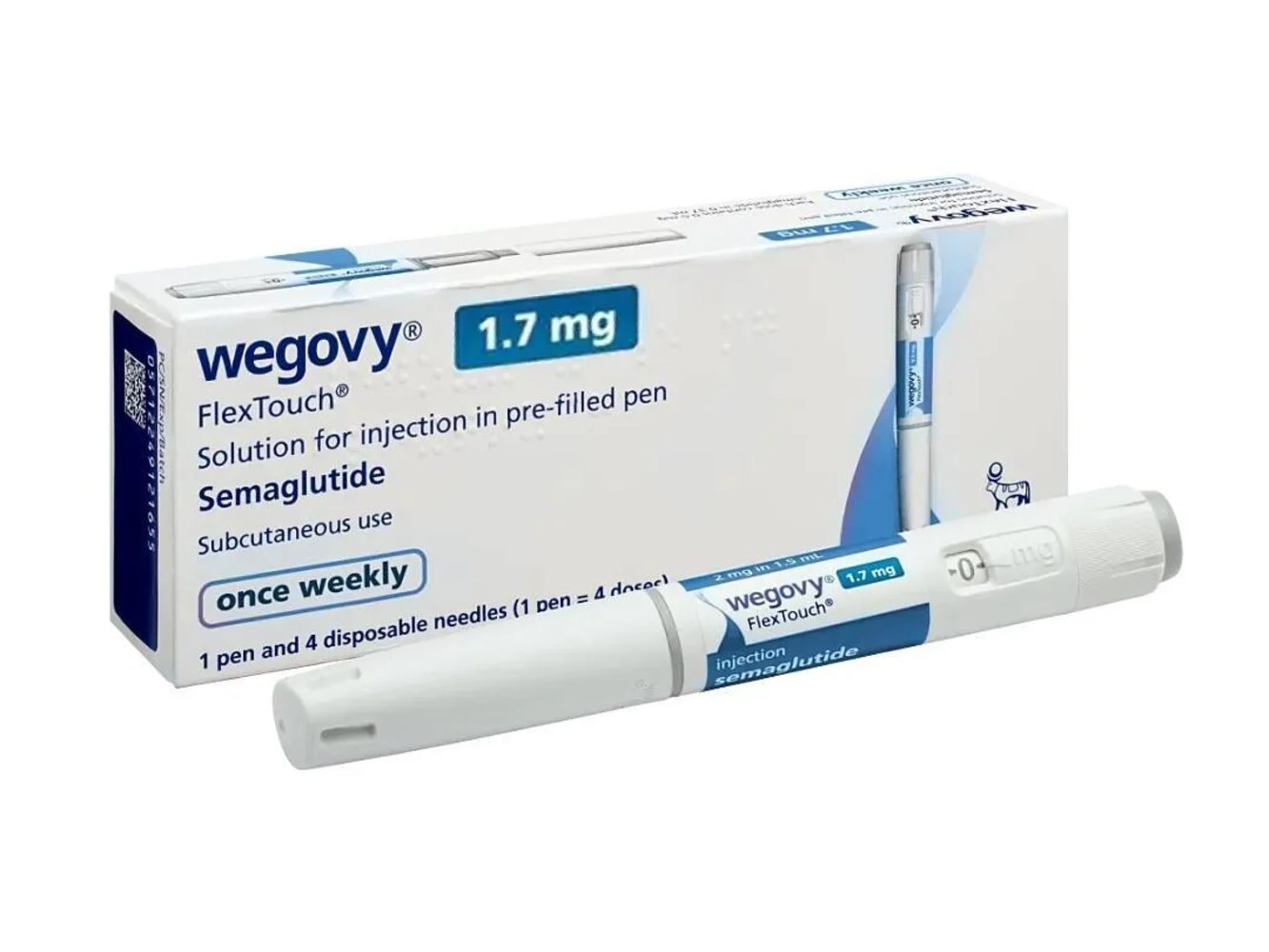
Wegovy 1.7 mg FlexTouch Pen (Semaglutide)
Price range: $620.00 through $1,760.00
| Active Ingredient: | Semaglutide |
|---|---|
| Indication: | weight loss and maintenance for overweight or obese adults |
| Manufacturer: | Novo Nordisk India |
| Packaging: | 1 Pen & 4 Disposable Needles |
| Strength: | 1.7mg |
| Delivery Time: | 10 to 15 days |
Wegovy 1.7 mg FlexTouch Pen (Semaglutide) — A Complete Guide
Wegovy 1.7 mg is the fourth dose in the Wegovy treatment programme. By the time you reach this stage, you have completed twelve weeks on semaglutide — four weeks each at 0.25 mg, 0.5 mg, and 1 mg. Arriving at 1.7 mg is a significant moment in the treatment journey. You are now one step away from the full maintenance dose, and the effects of the medicine at this level are among the strongest that most patients will have experienced so far.
This dose is taken during weeks thirteen to sixteen of treatment. For many patients, the 1.7 mg phase is when weight loss becomes clearly visible and consistent. Appetite is noticeably reduced, food portions feel naturally smaller, and the patterns of hunger and fullness that once drove overeating have shifted in a meaningful and lasting way. The body has had three months to adjust to semaglutide, and by this point, most patients have developed a good tolerance to the medicine.
The Journey So Far and What Comes Next:-
Understanding where Wegovy 1.7 mg sits within the full treatment schedule puts this phase in context and helps set realistic expectations for what lies ahead.
Weeks 1 to 4 — 0.25 mg: The initiation dose. The body is introduced to semaglutide gently. No weight loss is expected at this stage.
Weeks 5 to 8 — 0.5 mg: Tolerance continues to build. Some patients notice very subtle appetite changes. Weight changes are minimal at this point.
Weeks 9 to 12 — 1 mg: The midpoint. Appetite suppression becomes genuinely noticeable for most patients. Early and consistent weight loss often begins during this phase.
Weeks 13 to 16 — 1.7 mg: A powerful penultimate step. Appetite reduction is significant for most patients. Weight loss becomes more consistent and visible. The body is being prepared for the full maintenance dose.
Week 17 onward — 2.4 mg: The full maintenance dose. Maximum clinical benefit is achieved and sustained for as long as treatment continues.
If side effects at the 1.7 mg level are difficult to manage, your doctor may recommend staying at this dose for an additional four weeks before stepping up to 2.4 mg.
What Makes the 1.7 mg Dose Unique?
Every dose in the Wegovy 1.7 mg schedule has a role, but the 1.7 mg dose occupies a particularly important position. It is the final preparatory step before the full maintenance dose, and it is also the dose at which the medicine transitions from building tolerance to delivering near-full therapeutic benefit.
At 0.25 mg and 0.5 mg, the body is adjusting. At 1 mg, the medicine begins to work in a way most patients can feel. At 1.7 mg, those effects deepen considerably. The appetite suppression is stronger, the feeling of fullness after meals lasts longer, food cravings are more significantly reduced, and for most patients weight loss has become a consistent weekly occurrence rather than an occasional surprise.
Many patients describe the 1.7 mg phase as the point where Wegovy starts to feel effortless in the best sense — not that weight loss requires no effort at all, but that the constant battle with hunger and craving that once made eating less feel so difficult begins to ease fundamentally. Choosing smaller portions, leaving food on the plate, and passing on snacks no longer requires the same mental effort it once did.
How Wegovy 1.7 mg Works?
Wegovy 1.7 mg works through the same core mechanisms at every dose, but the depth and consistency of these effects increase with each dose step.
Hypothalamic appetite regulation. Semaglutide acts on GLP-1 receptors in the hypothalamus — the brain region that governs hunger, fullness, and energy balance. At 1.7 mg, this action is near its maximum. Hunger signals before meals are significantly blunted, and satiety signals after eating are stronger and more sustained. Most patients on 1.7 mg find that they simply do not feel as hungry as they once did — and when they do eat, they feel satisfied with noticeably less food.
Prolonged gastric emptying. The stomach continues to empty more slowly than normal while on Wegovy, keeping food present in the stomach for longer after meals. This physical fullness reinforces the brain-based appetite suppression and further reduces the urge to eat between meals.
Metabolic regulation. Semaglutide continues to support blood sugar regulation by stimulating insulin release in response to elevated glucose and suppressing the liver’s release of stored glucose. For patients who have insulin resistance, prediabetes, or type 2 diabetes alongside their obesity, these metabolic benefits become increasingly significant at higher doses.
Reduction in reward-driven eating. Semaglutide’s influence on the brain’s dopamine and reward pathways — which are strongly linked to food cravings and emotional eating — continues to deepen at 1.7 mg. Many patients report that the pull toward high-calorie comfort foods feels significantly weaker at this stage compared to when they first started treatment.
Increased energy expenditure. Emerging research suggests that semaglutide may have modest effects on the body’s resting energy expenditure — the number of calories burned at rest. While this is not the primary mechanism of weight loss, it may contribute to the overall calorie deficit that drives fat loss.
How to Use the Wegovy 1.7 mg FlexTouch Pen?
Wegovy 1.7 mg dose is used in the same way as the pens used throughout the earlier stages of treatment. It is pre-filled with a single weekly dose and is for one-time use only.
Approved injection sites. Wegovy can be injected into the abdomen, the front of the thigh, or the outer upper arm. For abdominal injections, always stay at least 2 inches away from the navel. It is essential to rotate the injection site each week to avoid skin changes, lumps, or irritation at the same spot.
Step-by-step injection guide:
- Wash your hands thoroughly with soap and water
- Remove the pen from the refrigerator a few minutes before use if you prefer a comfortable injection temperature
- Always check the label to confirm you are using the 1.7 mg pen — this is especially important as the pens look similar across dose strengths
- Remove the cap and inspect the medicine — it should appear clear and colorless with no particles
- Attach a fresh, sterile needle as directed in the package leaflet
- Clean the injection site with an alcohol swab if recommended by your healthcare provider
- Gently pinch the skin at the chosen site
- Press the pen firmly and evenly against the skin and push the injection button fully
- Hold the pen in place against the skin for a full six seconds to ensure complete delivery of the dose
- Remove the pen smoothly, release the pinched skin, and apply gentle pressure if needed
- Remove and safely discard the needle immediately in a sharps disposal container
- Discard the used pen — do not attempt to reuse it
Weekly injection schedule. Choose a consistent day of the week for your injection and stick to it. The injection can be taken at any time of day and does not need to be given with or without food. If you need to shift your injection day, ensure a minimum of two days have passed since your last dose before injecting the next one.
What to Realistically Expect at the 1.7 mg Stage?
Appetite and hunger. By the 1.7 mg phase, most patients experience a significant and consistent reduction in appetite. Hunger before meals feels less intense and more manageable. The feeling of fullness arrives sooner during meals and lasts longer afterward. Many patients find themselves eating noticeably smaller portions without having to consciously restrict themselves — the medicine is doing much of that work naturally.
Weight loss. For the majority of patients, weight loss is well established by the 1.7 mg phase and continues to progress. The rate of weight loss varies between individuals, but most patients on 1.7 mg are losing weight regularly and visibly. Some patients experience their most consistent and satisfying weight loss results during this phase, even before reaching the 2.4 mg maintenance dose.
Physical changes. Beyond the number on the scale, many patients begin to notice physical changes during the 1.7 mg phase — clothes fitting differently, improved mobility, reduced breathlessness during activity, and better energy levels throughout the day. These changes reinforce motivation and make the remaining steps of the treatment journey feel more meaningful.
Side effects. After twelve weeks on semaglutide, most patients have developed a good tolerance to the medicine. Side effects at the 1.7 mg dose are generally less disruptive than they were in the earlier phases, though some patients do experience a temporary return of nausea or digestive discomfort in the first one to two weeks after the dose increase. This is normal and typically settles quickly.
Mental and emotional changes. The psychological shift that many patients began noticing at 1 mg often deepens at 1.7 mg. Emotional eating, boredom eating, and habitual snacking become easier to resist. Food still brings pleasure, but the compulsive quality that often accompanies overeating in people with obesity begins to diminish. Many patients describe this as one of the most transformative aspects of their Wegovy experience.
Common Side Effects:-
Side effects at the 1.7 mg dose are generally consistent with those at earlier dose levels. For patients who have been tolerating the medicine well, side effects at this stage are often milder than in the early weeks of treatment. The most commonly reported include:
- Nausea — may return temporarily after the dose increase but usually settles within one to two weeks
- Diarrhea
- Constipation
- Vomiting
- Stomach pain or cramping
- Indigestion or heartburn
- Burping and bloating
- Fatigue during the dose adjustment period
- Reduced appetite — an intended therapeutic effect rather than a side effect in the traditional sense
To manage digestive side effects, continue eating smaller and more frequent meals, chew food slowly, avoid very fatty or spicy dishes, and maintain good hydration throughout the day.
Serious Side Effects:-
Pancreatitis. Sudden, severe pain in the abdomen or back that does not resolve may indicate acute pancreatitis. This is a medical emergency. Stop using Wegovy and seek immediate medical attention.
Gallbladder disease. The risk of gallstones and gallbladder problems remains present, particularly as weight loss accelerates. Symptoms include sharp pain in the upper right abdomen, fever, nausea, and jaundice. Inform your doctor without delay if any of these occur.
Thyroid tumors. The black box warning on Wegovy regarding the potential risk of thyroid C-cell tumors applies at all dose levels including 1.7 mg. Do not use Wegovy if you have a personal or family history of medullary thyroid carcinoma or Multiple Endocrine Neoplasia syndrome type 2.
Hypoglycemia. Patients also taking insulin or sulfonylureas remain at increased risk of low blood sugar. Monitor blood sugar levels regularly and contact your doctor if episodes of hypoglycemia become more frequent as the dose increases.
Heart rate changes. A mild elevation in resting heart rate has been observed in some patients using semaglutide. This is generally well tolerated but should be monitored, especially in patients with existing heart conditions.
Mental health changes. If you notice changes in your mood, increased feelings of anxiety or depression, or any thoughts of self-harm, contact your doctor immediately. These symptoms should always be taken seriously and discussed with a healthcare professional.
Kidney complications. If vomiting or diarrhea becomes prolonged and you are unable to stay adequately hydrated, kidney function can be affected. Contact your doctor if you cannot keep fluids down for more than a day or two.
Who Should Not Use Wegovy 1.7 mg?
- People with a personal or family history of medullary thyroid carcinoma
- Those diagnosed with Multiple Endocrine Neoplasia syndrome type 2
- Pregnant women or those planning pregnancy — Wegovy should be discontinued at least two months before attempting to conceive
- Women who are breastfeeding
- Anyone with a known hypersensitivity or allergy to semaglutide or any excipient in the injection
- People who have previously experienced a serious allergic reaction to any GLP-1 receptor agonist
Storage Instructions:-
- Store all unused Wegovy 1.7 mg FlexTouch Pens in the refrigerator at 2°C to 8°C
- Never allow pens to freeze — a pen exposed to freezing temperatures must be discarded
- Once removed from the refrigerator, a pen may be kept at room temperature below 30°C for up to 28 days
- Keep pens away from direct sunlight, heat, and damp conditions
- Store all pens in a location out of reach of children and pets
- Always check the expiry date before use and discard any pen that has passed its expiry date
Frequently Asked Questions (FAQs)?
Q1. What is Wegovy 1.7 mg used for?
Wegovy 1.7 mg is the fourth step in the five-stage dose escalation schedule. It is taken during weeks thirteen to sixteen of treatment and serves as the final preparatory dose before the full 2.4 mg maintenance dose. At this level, semaglutide delivers near-full appetite suppression and consistent weight loss for most patients. It is used as part of a comprehensive weight management programme that includes a reduced-calorie diet and increased physical activity.
Q2. How is 1.7 mg different from the earlier doses?
The 1.7 mg dose delivers a significantly stronger appetite-suppressing effect than the earlier doses. While the 0.25 mg and 0.5 mg doses are primarily about tolerance building, and the 1 mg dose marks the beginning of active therapeutic effects, the 1.7 mg dose brings patients very close to the full clinical benefit of Wegovy. Most patients notice a clear and consistent reduction in hunger, smaller natural portion sizes, and more regular weight loss at this stage.
Q3. Will my side effects get worse with Wegovy 1.7 mg?
Some patients experience a temporary return or mild worsening of nausea and digestive discomfort when stepping up to 1.7 mg, as the dose has increased from 1 mg. However, because the body has already had twelve weeks to adjust to semaglutide, most patients find that side effects at this stage are less severe and shorter-lived than in the early weeks of treatment. They typically settle within one to two weeks of the dose increase.
Q4. How much weight should I expect to lose at this stage?
Weight loss varies significantly between individuals and depends on factors including diet, physical activity, starting weight, and individual response to the medicine. By the 1.7 mg phase, many patients have already lost a meaningful percentage of their starting body weight. Weight loss typically continues to progress steadily at 1.7 mg and accelerates further once the 2.4 mg maintenance dose is reached.
Q5. Can I stay on Wegovy 1.7 mg permanently instead of going up to 2.4 mg?
This is a decision that must be made with your doctor. The recommended maintenance dose for Wegovy is 2.4 mg, as this is where the maximum clinical benefit is achieved. However, if you cannot tolerate the 2.4 mg dose due to persistent side effects, your doctor may consider keeping you at 1.7 mg as a long-term dose if it is producing adequate results for your individual situation.
Q6. I feel very little appetite at Wegovy 1.7 mg. Is this normal?
Yes, significantly reduced appetite is a normal effect of semaglutide at this dose level. Many patients on 1.7 mg describe having very little interest in food and needing to remind themselves to eat. While this supports weight loss, it is important to ensure you are still consuming enough nutrients each day. Focus on nutrient-dense foods when you do eat, and speak with your doctor or a dietitian if you are concerned about your food intake.
Q7. My weight loss has slowed down to Wegovy 1.7 mg. Should I be worried?
Weight loss naturally slows as the body adjusts to a lower calorie intake and a lower body weight. This is a normal physiological response and does not mean the medicine has stopped working. Continuing to 2.4 mg, maintaining a healthy diet, and staying physically active will support continued progress. Discuss any concerns with your doctor.
Disclaimer:-
The information provided in this article is for educational and informational purposes only. It is not intended to serve as a substitute for professional medical advice, diagnosis, or treatment. Always consult your doctor, pharmacist, or a qualified healthcare professional before starting, stopping, or making any changes to your medication or treatment plan. Never ignore or delay seeking professional medical advice based on anything you have read in this article. Results from Wegovy can vary from person to person, and only a licensed healthcare provider who is familiar with your complete medical history can determine whether this medication is appropriate for you. This article does not promote or endorse the use of any specific medicine. All medication decisions should be made in consultation with a qualified medical professional.
Similar Brands:-
Additional information
| mg | 1.7 Mg |
|---|---|
| unit | 1 Pen, 2 Pens, 3 Pens |




Reviews
There are no reviews yet.